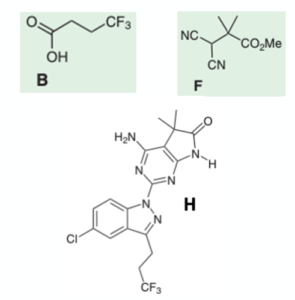
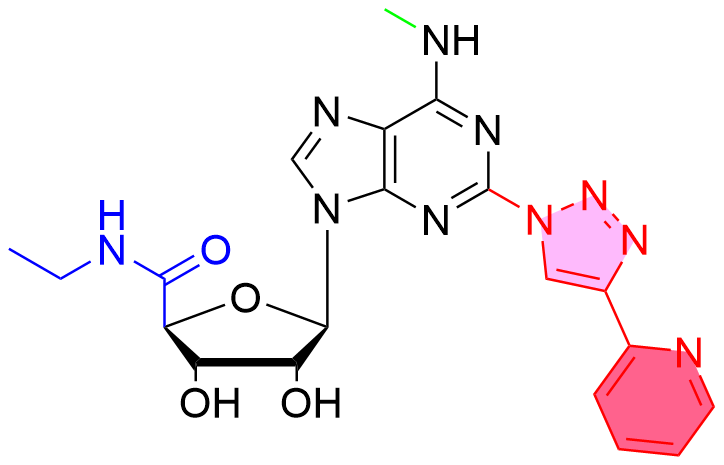
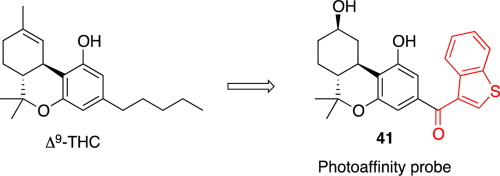
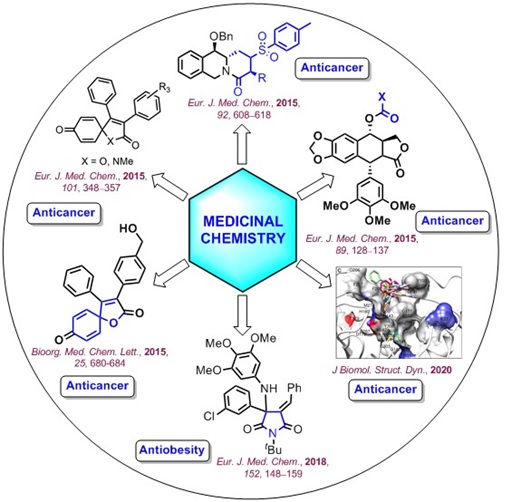
Bioorganic & Medicinal Chemistry Letters 1992: Vol 2 Index : Free Download, Borrow, and Streaming : Internet Archive
![PDF) Corrigendum to “Discovery and SAR of 2-amino-5-[(thiomethyl)aryl]thiazoles as potent and selective Itk inhibitors” [Bioorg. Med. Chem. Lett. 16 (2006) 2411–2415] | Tai-An Lin - Academia.edu PDF) Corrigendum to “Discovery and SAR of 2-amino-5-[(thiomethyl)aryl]thiazoles as potent and selective Itk inhibitors” [Bioorg. Med. Chem. Lett. 16 (2006) 2411–2415] | Tai-An Lin - Academia.edu](https://0.academia-photos.com/attachment_thumbnails/52287160/mini_magick20190123-29662-11w8dly.png?1548247061)
PDF) Corrigendum to “Discovery and SAR of 2-amino-5-[(thiomethyl)aryl]thiazoles as potent and selective Itk inhibitors” [Bioorg. Med. Chem. Lett. 16 (2006) 2411–2415] | Tai-An Lin - Academia.edu

Bioorganic & Medicinal Chemistry Letters | Vol 21, Issue 21, Pages 6207-6614 (1 November 2011) | ScienceDirect.com by Elsevier

Figure 3, Summary of second generation small molecule 3CLpro inhibitors, some containing reactive warhead groups and known to have a covalent mode of action - Probe Reports from the NIH Molecular Libraries

Bioorganic & Medicinal Chemistry Letters | Vol 26, Issue 15, Pages 3381-3830 (1 August 2016) | ScienceDirect.com by Elsevier
![PDF) ChemInform Abstract: Synthesis and in vitro and in vivo Anticancer Activity of Novel 3-Methyl-5H-isoxazolo[5′,4′:5,6]pyrido[2,3-b]indoles. PDF) ChemInform Abstract: Synthesis and in vitro and in vivo Anticancer Activity of Novel 3-Methyl-5H-isoxazolo[5′,4′:5,6]pyrido[2,3-b]indoles.](https://i1.rgstatic.net/publication/231860956_ChemInform_Abstract_Synthesis_and_in_vitro_and_in_vivo_Anticancer_Activity_of_Novel_3-Methyl-5H-isoxazolo5'4'56pyrido23-bindoles/links/5afbe176aca272e7302c496c/largepreview.png)
PDF) ChemInform Abstract: Synthesis and in vitro and in vivo Anticancer Activity of Novel 3-Methyl-5H-isoxazolo[5′,4′:5,6]pyrido[2,3-b]indoles.

Bioorganic & Medicinal Chemistry Letters | Recent Advances in Medicinal Chemistry | ScienceDirect.com by Elsevier

Molecules | Free Full-Text | Synthesis and Biological Evaluation of Novel Benodanil-Heterocyclic Carboxamide Hybrids as a Potential Succinate Dehydrogenase Inhibitors
![PDF) Erratum to “Rational design of novel, potent piperazinone and imidazolidinone BACE1 inhibitors” [Bioorg. Med. Chem. Lett. 18 (2008) 3236–3241] | Jared Cumming and Andrew Stamford - Academia.edu PDF) Erratum to “Rational design of novel, potent piperazinone and imidazolidinone BACE1 inhibitors” [Bioorg. Med. Chem. Lett. 18 (2008) 3236–3241] | Jared Cumming and Andrew Stamford - Academia.edu](https://0.academia-photos.com/attachment_thumbnails/41945149/mini_magick20190218-1404-1ork3dd.png?1550511543)